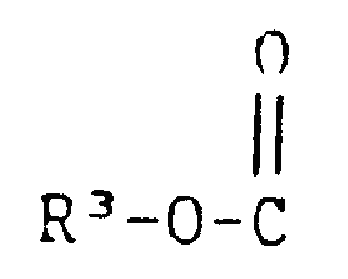Board of Patent Appeals and Interferences
Patent and Trademark Office (P.T.O.)
*1 DE SOLMS
v.
SCHOENWALD ET AL.
Patent Interference No. 101,698
February 22, 1990
Final Hearing: July 25, 1989
6-Amino-2-Benzothiazolesulfonamide and Topical Treatment Compositions and
Method for Glaucoma
Patent granted to S. Jane De Solms, Patent No. 4,499,103 issued February 12, 1985, filed March 17, 1983, Serial No. 476,002.
Application of Ronald D. Schoenwald and Charles F. Barfknecht filed April 4, 1985, Serial No. 720,006. Accorded Benefit of Serial No. 464,063, filed February 4, 1983.
William H. Nicholson, Mario A. Monaco, Rudolph J. Anderson, Jr. and Raymond M. Speer for De Solms. Oral argument by Raymond M. Speer
Donald H. Zarley, Bruce W. McKee, Dennis L. Thomte, Michael G. Voorhees, Edmund J. Sease, John A. Beehner, Mark D. Hansing and Kirk M. Hartung for Schoenwald et al. Oral argument by Edmund J. Sease
Before R. Smith, Sofocleous and Kimlin [FN1]
Examiners-in-Chief
R. Smith
Examiner-in-Chief
This interference involves a patent of De Solms, Patent No. 4,499,103, issued February 12, 1985 and assigned to Merck & Co., Inc., and an application of Schoenwald et al (Schoenwald), Serial No. 720,006, filed April 4, 1985 and assigned to University of Iowa Research Foundation.
The subject matter of the interference relates to benzothiazole-2-sulfonamide compounds which are useful for the topical treatment of elevated intraocular pressure. Count 1, the sole count at issue, reads as follows:
A compound of structural formula:
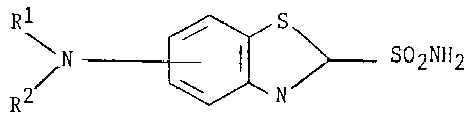
or an ophthalmologically acceptable salt thereof, wherein:
R1 and R2 are independently:
(1) hydrogen,
(2) C1-18 alkyl,
(3) C3-6 cycloalkyl,
(4) C3-6 cycloalkyl - C1-3 alkyl,
(5) aryl-C1-3 alkyl, wherein aryl is phenyl, naphthyl, pyridinyl, furanyl or thienyl, and is unsubstituted or substituted with one or more of chloro, bromo, fluoro, C1-3 alkyl or C1-3 alkoxy,
(6)
wherein R3 is
(b) aryl, either unsubstituted or substituted with one or more of chloro, bromo, fluoro, C1-3 alkyl or C1-3 alkoxy,
(c) aryl-C1-3 alkyl wherein the aryl group is either unsubstituted or substituted with one or more of chloro, bromo, fluoro, C1-3 alkyl, or C1-3 alkoxy,
(d) -amino-C1-18 alkyl; or
(7) R1 and R2, if loweralkyl, are joined together directly or through a heteroatom selected from O or N, to form a heterocycle with the nitrogen to which they are attached selected from pyrrolidine, piperidine, morpholine and piperazine.
The claims of the parties which correspond to this count are:
De Solms :Claims 1-16.
Schoenwald :Claims 1-14.
Schoenwald is senior party by virtue of the February 4, 1983 filing date of his parent application Serial No. 464,063. Schoenwald was accorded the benefit of the February 4, 1983 filing date by the primary examiner upon declaration of the interference. De Solms has not contested Schoenwald's entitlement to the benefit of his February 4, 1983 filing date during the motion period, nor in his brief at final hearing.
*2 As noted by De Solms in her brief and in accordance with the settlement agreement of March 15, 1988 between the parties, as interpreted by the Examiner-in-Chief in Paper No. 38, the issues at final hearing are limited to the priority issues of conception, diligence and reduction to practice.
De Solms' Case for Priority
De Solms, as the junior party to this interference, has the burden of proving prior invention by a preponderance of the evidence. Cf. Peeler v. Miller, 535 F.2d 647, 190 USPQ 117 (CCPA 1976). In an effort to prove prior invention De Solms submitted an evidentiary record including the declaration testimony of inventor De Solms and corroborating witnesses Christy, Schwam, Sugrue, Smith and Sondey and associated exhibits. References to the De Solms record will be designated as DR, followed by the record page number, and references to De Solms exhibits will be designated DX, followed by the declarant's name and exhibit number.
Dr. De Solms testified that she conceived of a new class of benzothiazole-2- sulfonamides as carbonic anhydrase enzyme inhibitors on or before September 17, 1981. Her conception included a suitable method of preparing the 6-amino compound by reduction of the nitro precursor (DR 13,14; DX De Solms 3). She began preparing the 6-amino compound on September 17, 1981, and by September 25, 1981 she established that the desired 6-amino-benzothiazole-2-sulfonamide had been prepared based on elemental analysis, proton nmr, carbon 13 nmr and HPLC (DR 15, 16, DX DeSolms 3, 4).
The compound prepared by De Solms was designated L-646,465-00F01 and was submitted for evaluation of its activity in inhibiting the carbonic anhydrase enzyme (DR 16). On October 23, 1981 Sondey completed the evaluation, and he testified that the results showed that compound L-646,465-00F01 "was an active inhibitor of the carbonic anhydrase enzyme"' (DR 37, 38, DX Sondey 4). De Solms urges that the successful demonstration of the ability of the 6-amino-benzothiazole-2-sulfonamide species within the genus of count 1 to inhibit the carbonic anhydrase enzyme establishes that an actual reduction to practice of the invention of the count took place on October 23, 1981.
Opinion re De Solms' Case for Priority
We hold that De Solms has established conception of the invention of count 1 by September 17, 1981, when De Solms began the preparation of 6-amino-benzothiazole-2-sulfonamide, as corroborated by Christy and Smith. We hold further that De Solms has proven by a preponderance of the evidence actual reduction to practice of the invention of count 1 by October 23, 1981, prior to Schoenwald's February 4, 1983 effective filing date.
Schoenwald's argument that there is no corroboration of Sondey's work in evaluating compound L-646,465-00F01 for carbonic anhydrase enzyme inhibition activity is not persuasive. Sondey is not an inventor, and it is well settled that the purpose of the corroboration requirement is to verify the testimony of the inventor. We reject Schoenwald's argument, that the Sondey's testimony must be corroborated even though he is not an inventor, as contrary to interference law. See III Rivise and Caesar, Interference Law and Practice, § 539 (Michie Co. 1947) and the cases cited therein, for the proposition set forth on page 2128 as follows:
*3 It is important to note that the requirement of corroboration applies only to the testimony of a person claiming to be an inventor. If the inventor was assisted in his work, the assistant's testimony as to what he did need not be corroborated.
Further, see Klug v. Wood, 212 USPQ 767 (Bd. Pat. Int. 1981) where the Board held that "[s]ince Stigen was not an inventor, no corroboration of his work is necessary."D'
Similarly, Schoenwald's argument that Sondey does not state that he showed or sent the information to anyone and De Solms does not indicate she ever received it is not persuasive. In our view it is unnecessary for the test results to be communicated to De Solms to establish a reduction to practice. Since De Solms had a conception of the invention by her preparation and identification of the compound coupled with her expectation of carbonic anhydrase enzyme inhibiting activity, the work of Sondey in evaluating the compound accrued to De Solms' benefit whether she knew about it or not. Little v. Cowling, 26 CCPA 874, 101 F.2d 541, 40 USPQ 492 (1939); Gunter v. Stream, 573 F.2d 77, 197 USPQ 482 (CCPA 1978). The evidence shows that Sondey was an employee of Merck, that he reduced the invention to practice in the course of his employment and that he has not contested the designation of De Solms as the inventor (DR 23-25, 36- 38). The evidence satisfies us that the reduction to practice was on De Solms' behalf by persons authorized to do so. Cf. Haskell v. Colebourne, 671 F.2d 1362, 213 USPQ 192 (CCPA 1982).
Schoenwald's Case for Priority
Schoenwald, the senior party, has presented an evidentiary record including the declaration testimony of co-inventors Barfknecht and Schoenwald, both professors of pharmacy at the University of Iowa, the declaration testimony of their graduate students, Dixson and Eller, and associated exhibits. References to the Schoenwald record will be designated as SR, and references to the Schoenwald exhibits will be designated SX, followed by the exhibit number. Schoenwald relies on the evidence in an effort to establish conception of the invention of the count prior to the September 17, 1981 conception accorded to De Solms, supra, and either actual reduction to practice on October 7, 1981, when a compound within the count was prepared, or reasonable diligence from prior to De Solms' September 17, 1981 entry into the field until Schoenwald's constructive reduction to practice on February 3, 1983.
Dixson testified that he was a graduate student working under the direction of Professor Barfknecht, and that his assignment involved the preparation of compounds to be investigated as possible carbonic anhydrase inhibitors (SR 44- 46). One of the compounds known as D-3 was 6-amino-2-benzothiazolesulfonamide and another known as D-5 was 6-hydroxyethoxy-2-benzothiazolesulfonamide. Dixson testified further that his efforts to synthesize compounds D-3 and D-5 began in the summer of 1981 (SR 46). From the summer of 1981 through February 4, 1983 Dixson worked on the synthesis of many different 2-benzothiazolesulfonamides, "at least 17 benzothiazole final products"' were prepared, and with intermediates required the synthesis of more than 60 compounds (SR 46-47). Dixson synthesized compound D-3 and verified its structure by October 7, 1982 (SR 49; SX 20, 21). On November 15, 1982 he submitted the compound D-3 to Schoenwald for further work by his graduate student, Eller (SR 6; SX 23).
*4 Eller testified that he was a graduate student working for Dr. Schoenwald on projects in the area of carbonic anhydrase inhibitors (SR 51, 52). Eller worked "on various carbonic anhydrase inhibitors that included the parent or prototype structure of 2-benzothiazolesulfonamide" and investigated the "pharmaceutical activities of these compounds virtually continuously from June 1980 until I received my Ph.D. degree in 1984"' (SR 52). Eller's work included developing the physical and chemical properties of compounds prepared by Barfknecht and Dixson, determination of solubility, pKa and partitioning, responsibility for testing carbonic anhydrase inhibition by the Maren Micro Method, responsibility for corneal permeability testing, and intraocular pressure determinations in vivo, in rabbit eyes.
Opinion re Schoenwald's Case for Priority
We hold that Schoenwald has established by a preponderance of the evidence conception of the invention of count 1 by June 11, 1981, prior to the September 17, 1981 conception accorded De Solms, supra, and that Schoenwald has established reasonable diligence from prior to September 17, 1981 until his constructive reduction to practice on February 4, 1983.
With respect to conception, Schoenwald points out in his brief (page 23 and 24) that De Solms "admits Schoenwald conceived the invention prior to its own conception date,"' and De Solms has not contested, in his reply brief, nor at final hearing, Schoenwald's contention that prior conception by Schoenwald has been admitted. Rather, De Solms conceded at final hearing that Schoenwald has an earlier conception, i.e., by June 11, 1981.
Schoenwald contends at page 26 of his brief that
[I]f the Board believes that the utility of this compound was apparent because of its similar structure to other 2-benzothiazolesulfonamides having carbonic anhydrase inhibition properties, then synthesis of the compound on October 7, 1982 was an actual reduction to practice of the compound."D'
We believe that the situation here, like that in Fisher v. Bouzard, 3 USPQ2d 1677, 1680-81 (BPAI 1987), is analogous to that in Blicke v. Treves, 44 CCPA 753, 241 F.2d 718, 112 USPQ 472 (1957). Unless the utility could have been foretold with certainty, sufficient testing to establish the practical utility of the compound is required to establish an actual reduction to practice. Schoenwald points to no evidence that would persuade us that the utility of compound D-3 as a carbonic anhydrase inhibitor could have been foretold with certainty at the time it was prepared. Accordingly, we hold that Schoenwald has not established an actual reduction to practice of the count in issue by October 7, 1982, nor at any time prior to his constructive reduction to practice on February 4, 1983.
In order to prevail on the basis of prior conception coupled with reasonable diligence, Schoenwald must establish that he was reasonably diligent from a time just prior to De Solms' conception on September 17, 1981 until Schoenwald's constructive reduction to practice on February 4, 1983, a period of more than 16 months. After a careful consideration of all of the evidence presented by Schoenwald and the positions of the parties as set forth in their briefs, we have concluded that Schoenwald has established the requisite reasonable diligence.
*5 De Solms urges that in judging the reasonableness of Schoenwald's activities during the critical period it should be noted that De Solms was able to reduce the invention to practice in 35 days from the time that the preparation of the compound was begun. Similarly, De Solms notes that at the beginning of the diligence period seventeen 2-benzothiazolesulfonamide compounds had been proposed, and eight of these had already been made. It took Dixson, who was working only one-half time, as long as a month to make one compound, and it took until October 7, 1982 to make the 6-amino compound of the count, whereas De Solms made the same compound in 8 days. De Solms urges that it was a conscious decision on the part of Schoenwald's assignee to require Schoenwald to seek outside support and to rely on a graduate student of limited experience which resulted in the delay, citing Griffith v. Kanamaru, 231 USPQ 892 (BPAI 1986).
We believe that De Solms' reliance on Griffith is misplaced. Griffith sought to excuse a gap in activity where there was no work toward a reduction to practice of the invention based on the need to secure outside funding for the project before the work could proceed. In contrast to the situation in Griffith, Schoenwald and Barfknecht had funding for their project throughout the diligence period, and it is clear from the testimony of Dixson and Eller that they were diligently working on the project to the extent that their positions as graduate students allowed throughout the critical period. It is a principle of diligence that consideration must be given to the circumstances of the inventor, including his skill and available time, and that the inventor may avail himself of the activities of others. I Rivise and Caesar, Interference Law and Practice, §§ 191,198 (Michie Co. 1940). Accordingly, we find that the diligent activities on behalf of Schoenwald are not negated by the fact that Dixson and Eller did not possess the same level of skill as De Solms or that Dixson and Eller were not able to work full time on their project. Similarly, it is not material to the question of diligence that Schoenwald did not take the most expeditious course, so long as there was diligent activity toward the end in view. I Rivise and Caesar, Interference Law and Practice, supra, § 196.
The central issue as to Schoenwald's case for diligence is whether Schoenwald is entitled to consideration of work by Dixson in preparing compounds outside the scope of the count. In this connection De Solms relies on the decision in Smith v. Crivello, 215 USPQ 446 (Bd. Pat. Int. 1982), for the proposition that a party charged with diligence may not rely on activities directed toward another independent invention. We find, however, that the facts of this case are more similar to those in Ginos v. Nedelec, 220 USPQ 831 (Bd. Pat. Int. 1983) than they are to Smith, supra. Whereas Smith was not permitted to rely on activities directed toward another independent invention, Ginos was continuously working on the preparation of other compounds which were considered by Ginos to be part of the same invention and which were included in his application. We believe that Schoenwald is similarly entitled to be credited with the work of Dixson in preparing compounds within the scope of Schoenwald's invention of 2-benzothiazolesulfonamide compounds which were included in Schoenwald's parent application and were manifestly considered by Schoenwald as part of the same invention.
*6 De Solms urges that the public policy favoring early disclosure of inventions should preclude according Schoenwald with a "reservoir of reasonable diligence"' on the basis of the ongoing carbonic anhydrase inhibitor research project which included much work on compounds outside the scope of the present count. De Solms' reliance on the public policy favoring early disclosure ignores the fact that Schoenwald was the first to file his patent application and was therefore the first to initiate the process leading to public disclosure. Thus, the public policy favoring early disclosure supports our decision according priority of invention to Schoenwald.
Decision
For the foregoing reasons, judgment as to the subject matter of count 1 is awarded to Ronald D. Schoenwald and Charles F. Barfknecht, the senior party. Accordingly, De Solms is not entitled to a patent containing claims 1-16, corresponding to the count, and Schoenwald is entitled to a patent containing claims 1-14, corresponding to the count.
BOARD OF PATENT APPEALS AND INTERFERENCES
Ronald H. Smith
Examiner-in-Chief
Michael Sofocleous
Examiner-in-Chief
Edward C. Kimlin
Examiner-in-Chief
FN1. Examiner-in-Chief Kimlin has been substituted for Examiner-in-Chief Rollins, who has retired.
<< Return to Board of Patent Appeals and Interferences Index